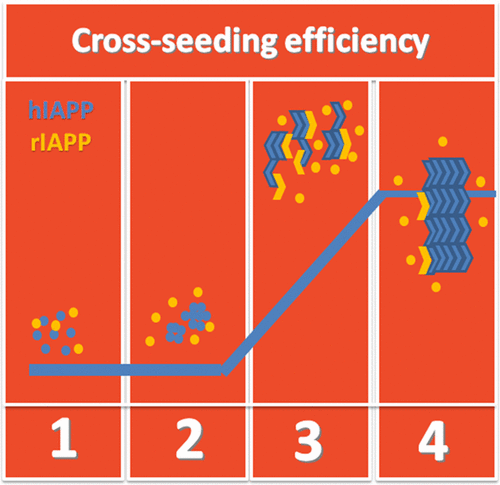
ABSTRACT:Amyloid peptides can misfold and aggregate into amyloid oligomers and fibrils containing conformationally similar β-sheet structures, which are linked to the pathological hallmark of many neurodegenerative diseases. These β-sheet-rich amyloid aggregates provide common structural motifs to accelerate amyloid formation by acting as seeds. However, little is known about how one amyloid peptide aggregation will affect another one (namely, cross-seeding). In this work, we studied the cross-seeding possibility and efficiency between rat islet amyloid polypeptide (rIAPP) and human islet amyloid polypeptide (hIAPP) solution with preformed aggregates at different aggregation phases, using a combination of different biophysical techniques. hIAPP is a well-known peptide hormone that forms amyloid fibrils and induces cytotoxicity to β-cells in type 2 diabetes, whereas rIAPP is a nonaggregating and nontoxic peptide. Experimental results showed that all different preformed hIAPP aggregates can cross-seed rIAPP to promote the final fibril formation but exhibit different cross-seeding efficiencies. Evidently, hIAPP seeds preformed at a growth phase show the strongest cross-seeding potential to rIAPP, which accelerates the conformational transition from random structures to β-sheet and the aggregation process at the fibrillization stage. Homoseeding of hIAPP is more efficient in initiating and promoting aggregation than cross-seeding of hIAPP and rIAPP. Moreover, the cross-seeding of rIAPP with hIAPP at the lag phase also reduced cell viability, probably because of the formation of more toxic hybrid oligomers at the prolonged lag phase. The cross-seeding effects in this work may add new insights into the mechanistic understanding of the aggregation and coaggregation of amyloid peptides linked to different neurodegenerative diseases